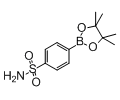
4-Sulphamoylbenzeneboronic acid, pinacol ester
4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzenesulfonamide
CAS: 214360-51-7
Molecular Formula: C12H18BNO4S
4-Sulphamoylbenzeneboronic acid, pinacol ester - Names and Identifiers
4-Sulphamoylbenzeneboronic acid, pinacol ester - Physico-chemical Properties
| Molecular Formula | C12H18BNO4S |
| Molar Mass | 283.15 |
| Density | 1.24±0.1 g/cm3(Predicted) |
| Boling Point | 427.1±47.0 °C(Predicted) |
| Flash Point | 212.129°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 10.00±0.10(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.543 |
4-Sulphamoylbenzeneboronic acid, pinacol ester - Reference Information
| Application | Phenylsulfonamido-4-boric acid is a boric acid derivative, boric acid derivatives are widely used in organic synthesis for the formation of carbon-carbon bonds. In Suzuki coupling, aryl halides and boric acid aryl or vinyl ester or boric acid are coupled using Pd(PPh3)4. |
| Uses | Phenylsulfonamido-4-boric acid is a carboxylic acid derivative, mainly used as a pharmaceutical intermediate. |
| preparation | potassium acetate (2.5g, 24.4 mmol) is added to a solution of 4-bromobenzenesulfonamide (2.00g, 9.32 mmol) and bis (pinacol) diboron (2.40g, 9.32 mmol) in DMSO(20 ml) and degas the mixture for 45 minutes. Then [1,1 '-bis (diphenylphosphino) ferrocene] dichloropalladium (II)(220 mg, 0.26 mmol) was added and the mixture was heated to 90°C for 16 hours. Once cooled, the reaction mixture was diluted with EtOAc(30 ml), washed with water (3 × 30 ml), dried with MgSO4 and concentrated under vacuum. The residue was ground from Et2O(50 ml) and HCl(1M,50 ml), the resulting solid was dissolved in CH2Cl2(30 ml) and filtered through a silicone pad cleaned with Et2O, and then concentrated to obtain phenylsulfonamido-4-boric acid as an off-white solid, with a 13% yield of 550 mg. |
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)benzenesulfonamide Visit Supplier Webpage Request for quotationCAS: 214360-51-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzenesulfonamide Request for quotation
CAS: 214360-51-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 214360-51-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4-SULFAMOYLPHENYLBORONIC ACID, PINACOL ESTER Request for quotation
CAS: 214360-51-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 214360-51-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 4-Sulfamoylphenylboronic Acid Pinacol Ester Visit Supplier Webpage Request for quotation
CAS: 214360-51-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 214360-51-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)benzenesulfonamide Visit Supplier Webpage Request for quotationCAS: 214360-51-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzenesulfonamide Request for quotation
CAS: 214360-51-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 214360-51-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4-SULFAMOYLPHENYLBORONIC ACID, PINACOL ESTER Request for quotation
CAS: 214360-51-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 214360-51-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 4-Sulfamoylphenylboronic Acid Pinacol Ester Visit Supplier Webpage Request for quotation
CAS: 214360-51-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 214360-51-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



